We follow IS 10500:2012 for drinking water standards. The properties/parameters which help in asserting the quality of raw water are called Water Quality Parameters.
These parameters are classified as :-
1.Physical Water Quality Parameters
2.Chemical Water Quality Parameters (Click on it)
3.Biological Water Quality Parameters ( Click on it)
Note – There are 2 limits usually associated with these parameters namely Acceptable Limit and Cause for Rejection.
PHYSICAL WATER QUALITY PARAMETERS
The parameters which can be felt or sensed by any of our senses are referred as physical water quality parameters which are mentioned below:
- Suspended Solids
- Turbidity
- Colour
- Taste And Odour
- Temperature
Types of Impurities in Water
There are two types of impurities – Organic and Inorganic. Organic compounds always composed of carbon products while most inorganic compounds do not contain carbon.
Examples –
- Organic Compounds : nucleic acids, fats, sugars, proteins, enzymes, hydrocarbons, methane etc.
- Inorganic Compounds : salts, metals, substances made from single elements and compounds which don't contain carbon bonded to hydrogen etc.
Almost all organic compounds contain carbon-hydrogen or C- H bonds except compounds like CCl4, NH2-CO-NH2 etc.
Note : Containing carbon is not sufficient but necessary condition for a compound to be considered organic.
1.TOTAL SOLIDS
- Total Solids quantify all the solids present in water i.e. suspended and dissolved, organic and inorganic.
- Measured by gravimetric method i.e. by weighing
- Found out by evaporating the test sample at 104ᵒC. The weight of dry residue is Total Solid.
- Represented in mg/l usually. (1 mg/l = 1 ppm (only for water))
- Limits
Acceptable limit : 500 mg/l
Cause for rejection limit : 2000 mg/l
SUSPENDED SOLIDS (SS/TSS)
Sources : Suspended Solids in water comes from inorganic particle like silt, clay etc., organic particles like plant fibers, algae etc.
Measurement: Measurement is done by graviometric technique i.e. weighing.
- Total Solids(TS) = SS + DS are calculated by evaporating the sample at 104ᵒC and measuring the residue.
- SS is obtained by filtration and heating the residue on filter at 104ᵒC .
- TDS = TS – TSS
- Suspended Solids are also called as Non-Filterable Solids
- Dissolved Solids are also called as Filterable Solids
- Organic solids both in total and suspended form can be determined by firing the test sample and residue in muffle furnace at 600-650ᵒC.
- At this temp organic solids will be vaporized leaving behind inorganic solids.
- Organic fraction @600ᵒC gives Carbon dioxide + water + gases
- Organics solids are also called as volatile solids and Inorganic solids are also called as fixed solids.
- There must not be any suspended solids in the drinking water.
2. TURBIDITY
Turbidity is the measurement of extent by which light is either absorbed or scattered by suspended material in water. However it is not a direct quantitative measure of Suspended Solids.
Note – Suspended solids depends upon concentration whereas turbidity depends on both concentration and fineness of particles in water.
Impacts :
- Disinfection of turbid water is difficult because the SS may partially shield the organisms from disinfectant.
- In natural bodies, turbidity interferes with light penetration and hence with the photosynthetic reactions (which gives oxygen to the water).
Note – Turbidimeter works on the principle of measuring the interference caused by a water sample to the passage of light rays.
Limits :
Acceptable Limit : 1 NTU
Cause for Rejection : 5 NTU
3. COLOUR
Source: Colour is caused by suspended and dissolved matter in water referred as apparent colour.
After suspended matter causing colour is removed by filtration, centrifugation etc., the colour obtained is called true colour.
Measurement: Measurement of colour is done by colour matching techniques using Nesslers Tubes ( Instrument used - Tintometer).
Result is expressed in TCU (True Colour Unit) or Hazen unit where 1 TCU is equal to colour produced by 1 mg per litre of platinum in the form of chloroplatinate ions(also called as
Burgess Scale). TCU is also called as Hazen Unit.
(It is only for yellowish brown colour).
The colour testing is done within 72hrs of collection as otherwise biological or physical properties may change.
Limit : Acceptable limit is 5 TCU
Cause for Rejection is 15 TCU (previously 25)
4.TASTE AND ODOUR
Source
Taste and odour are caused by dissolved gases like Hydrogen Sulphide, Methane, Organic matter or from inorganic matter.
Measurement
Odour is generally measured by an instrument known as Osmoscope by diluting the water sample upto an extent it is hardly detectable.
Intensity of taste and odour is represented and expressed as Threshold Odour Number (TON)
It represents the dilution ratio at which odour is hardly detectable
TON = (A + B) / A where A is the volume of odorous water in ml and B is the volume of odour free water required to produce a mixture in which odour is hardly detectable.
Increase in temperature may change the taste and odour. Due to this, TON testing is done in cold water.
Limit:
Acceptable limit : 1 TON
Cause for rejection : 3 TON
Note – The substances which impart odour also produce some taste but vice versa is not true.
5.TEMPERATURE
- Temperature affects the chemical and biological reactions.
- An increase in 10ᵒC almost doubles the biological activity.
- For water supply, the temperature should be between 10-25ᵒC








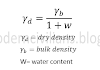



1 Comments
Tds water testing is very essential to know the quality of the water. A high TDS level may refer to the consumption of bad contaminants, that are salts and minerals or even metals. Regular tests ensure drinking water is safe and healthy. Use accurate TDS Water Testing to guarantee your water's purity.
ReplyDelete